Not everything is easy to see. Some things are actively trying to hide whilst others are just hard or impossible to see with the naked eye. What we would like is a tool that enhances our ability to see beyond the visible spectrum. Even better if we can recognise what it is we are looking at.
Thermal optics do that by allowing us to see into the infrared part of the spectrum. By treating everything like a black body, we can even estimate the temperature of different things that are being imaged and highlight things like humans for ease of recognition. Often to do that we sacrifice the visual spectrum or have to superimpose it with a fused view.
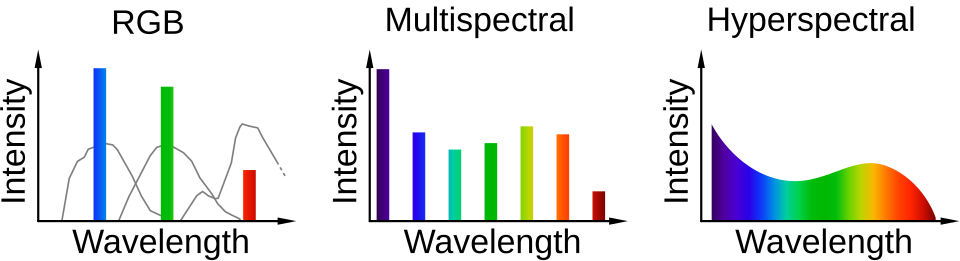
Hyperspectral imaging (HSI) goes several steps further. Where a normal camera would give you how much red, green, and blue are in each pixel in an image, and an IR camera might give you some channels in the infrared, HSI gives you detail into the whole spectrum of its range down to bands of a few nanometres.
This phenomenal amount of data means you have what is essentially an FTIR spectrum (like you would get from a commonly used TruDefender from Thermofisher) for each pixel in an image. Much like you would use an FTIR spectrum, that means you can start to understand the material of anything you image with a HSI system.
For the CBRNE world that means a few things. First, we might be able to use them to detect things that are camouflaged in the visible part of the spectrum. The famous Houthi roadside IEDs that were hidden as rocks would have a wildly different spectral fingerprint compared to a real rock. I have never scanned a rock, but it strikes me as the kind of thing with different functional groups to the polyurethane of expanding foam. The right kind of anomaly detection algorithm would make detecting those very easy.

The second that comes to mind is chemical detection. Right now we lack the tools to effectively recognise where gaseous chemicals have spread to without putting detectors in various places and estimating the gaps inbetween. With a hyperspectral camera it is straightforward to identify gases based on their hyperspectral fingerprint from a distance, with the usual proviso that they have IR-active groups in the bands being measured.
This would mean HAZMAT jobs with a gaseous contaminant have to do less guess work when it comes to how they are containing and venting things, or how far a particular gas has spread. Plotting a downwind hazard after a release goes from a guess to quantifiable amount with a UAS-mounted HSI.

The spectral fingerprinting I have described here is a feature of hyperspectral imaging. It is worth contrasting this with multispectral imaging which still gives some insight into the spectrum past the visible portion by showing bands of infrared. Much less study has gone into whether or not multispectral cameras are “good enough” for the purposes I have discussed above and it is worth a shot to investigate if it means more people can get access to seeing in infrared.
Next time, we will have a look at what imaging systems have been studied for the kinds of defence applications discussed here. Whilst I hope I have painted a hopeful picture for the technology, and a lot of work has been done to show its efficacy right now, for CBRNE there are still a lot of employment questions left to be tackled.





